Covid-19 vaccine takes a step forward in Seattle
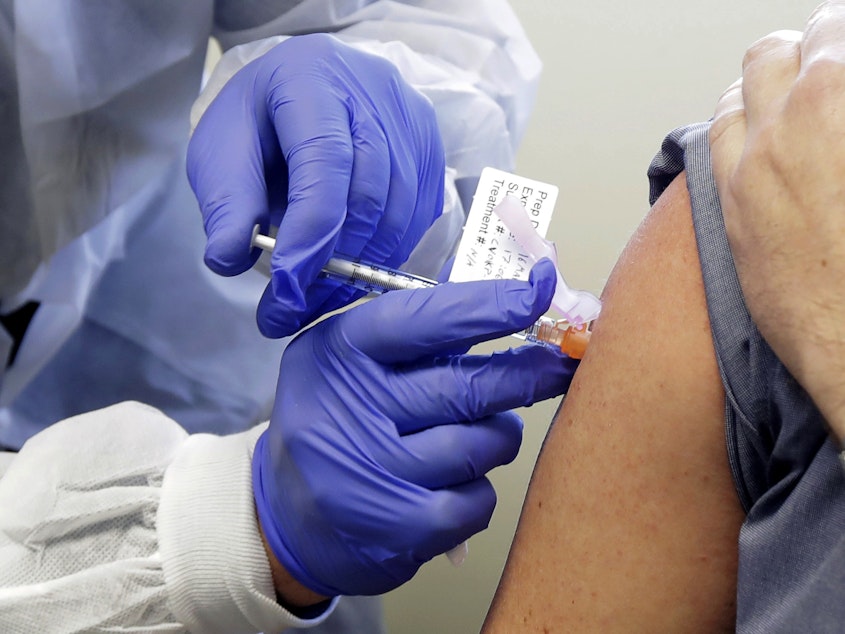
A Covid-19 vaccine tested in Seattle took another step forward Tuesday.
Moderna’s messenger-RNA vaccine is safe enough for clinical trials to continue. There are also signs it may work.
The preliminary safety trial (Phase 1) results were published Tuesday in the New England Journal of Medicine.
Volunteers injected with two doses of the vaccine at Kaiser Permanente Research Institute in Seattle developed antibodies comparable to someone who has had the disease.
3 people on the frontlines of developing a coronavirus vaccine
But it’s not yet clear how well this vaccine will work. There are still two more phases of clinical trials to go.
This is the first coronavirus vaccine to be tested on humans. And it could be the first vaccine publicly available if all goes well.
Sponsored
The lead author of the study is Lisa A. Jackson, MD, MPH, a senior investigator at Kaiser Permanente Washington Health Research Institute in Seattle.
An editorial in the New England Journal of Medicine notes that through this vaccine trial, "the world has now witnessed the compression of 6 years of work into 6 months."
"The speed with which this vaccine has been developed is remarkable — from publication of the first SARS-CoV-2 sequences through phase 1 in 6 months, as compared with a typical timeline of 3 to 9 years .... The safety and immunogenicity data in this preliminary report are promising, and they support continued development of this vaccine. However, we must bear in mind the complexity of vaccine development and the work still to be done before Covid-19 vaccines are widely available."
Read more on KUOW
Seattle man spikes fever after second dose of trial coronavirus vaccine
Sponsored
Checking in with the Seattle woman who took the first dose of an experimental coronavirus vaccine
New Coronavirus Vaccine Candidate Shows Promise In Early, Limited Trial




